We are specializing in Thermal Validation in the various fields like Pharmaceutical, Biotechnology, Medical Devices industries, Beverages etc.
Equipment Validation :
Equipment Validation ensures that an instrument is appropriate for its intended use. Typical validation phases are:
ü Double door / Vertical Autoclave.
ü Oven.
ü Dry heat Sterilizer.
ü Tray dryer / Vacuum Tray Dryer.
ü Tunnel Sterilizer.
ü Cold room BOD Incubator.
ü Room Study (Temp. & RH)
ü Water / Dry Bath.
ü Manufacturing vessel.
ü Refrigerator / Deep Freezer.
ü Muffle / Bunsen Furnace .
QTS is well equipped with 600+ portable loggers, 400 + sensors, 8 chartless Data loggers (12 To 50 channels).
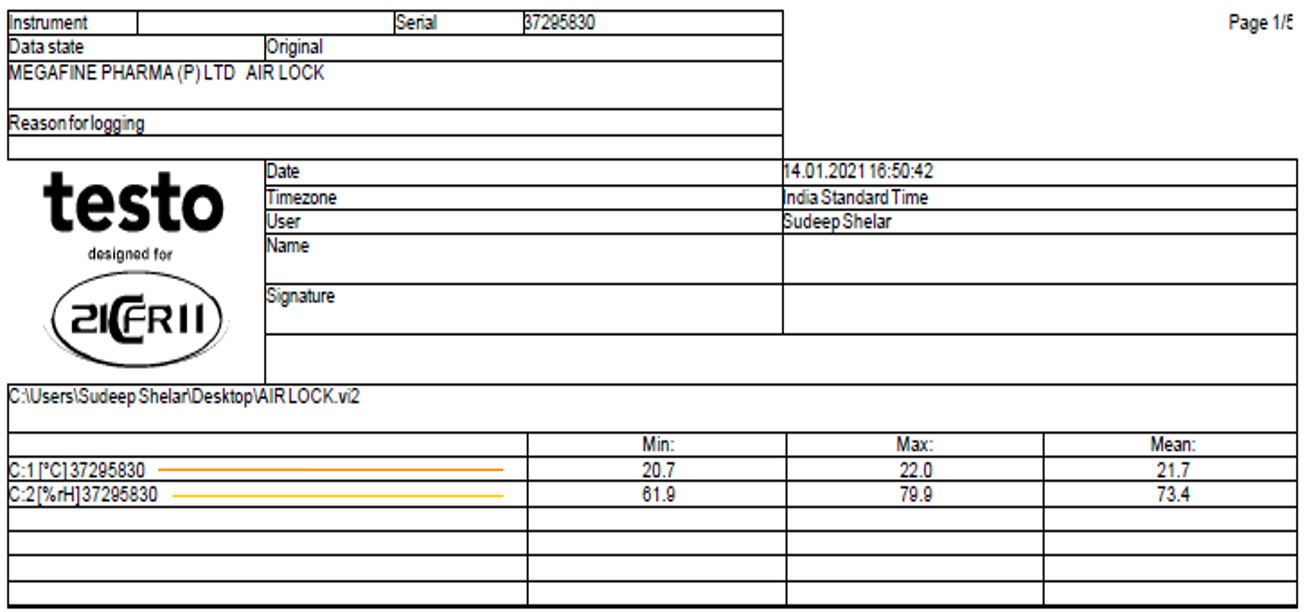
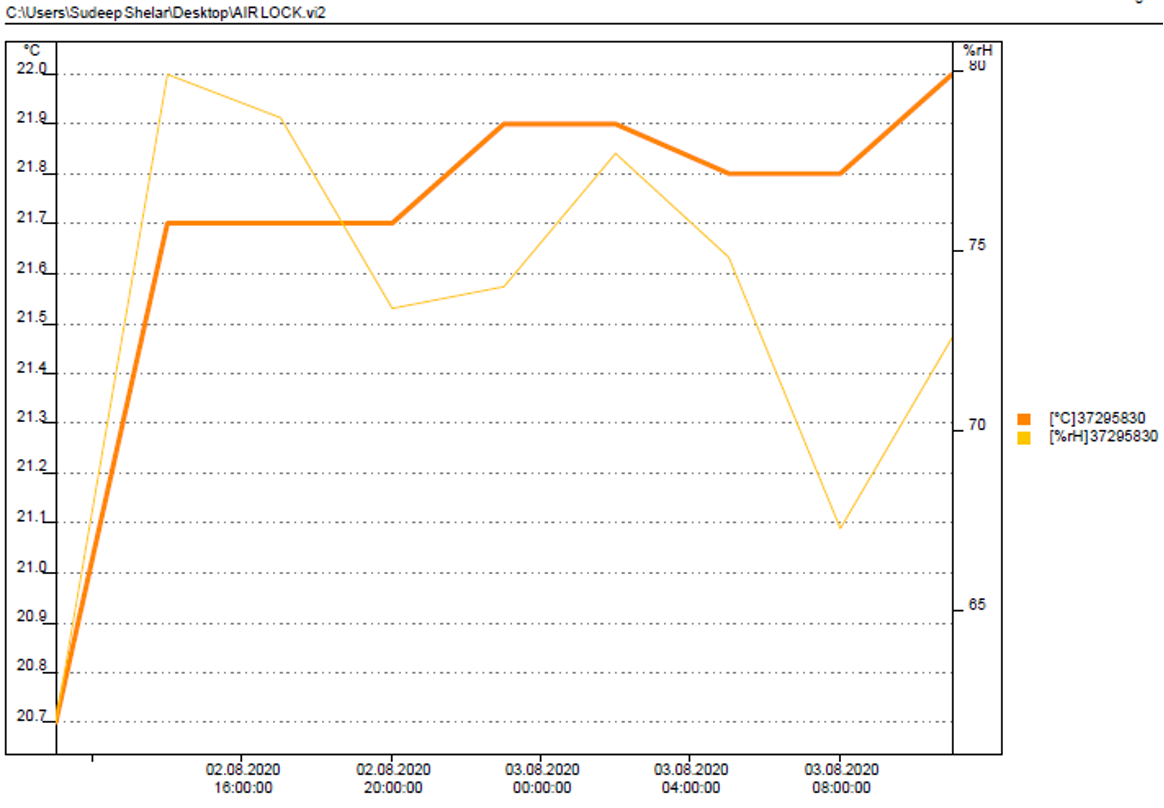
The data logged from the mapping of the above equipment is further processed to give analysis reports like F0 values, lethal rate, standard deviation, lag time in case of autoclaves and, Hot spot and cold spot identification in case of other equipments and rooms.
This process can be certified by 21 CFR Part 11.